The proton was discovered by Ernest Rutherford in 1919. Hence, the mass number (A) is the sum of both sub-particles.Īnother essential characteristic is that its positive charge helps capture electrons and keep them orbiting around the atom's nucleus. The number of neutrons (N) is also used by adding all the nucleons to determine the isotopes of an element. The atomic number (Z) is the number of protons in its nucleus and determines the atom's chemical properties. Protons are essential because they define what element an atom is. Therefore, neutral atoms have the number of electrons, and protons must be the same. Protons and electrons have the same absolute charge but with opposite signs. The proton has a positive elemental charge of 1.602 x 10 -19 coulombs. However, compared to the electron, the proton's mass is about 1,836 times greater. 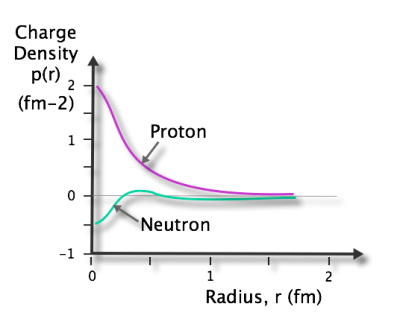
This value is roughly the same mass as neutrons. It has a mass of about 1.6726219 × 10 -27 kilograms. The laws of physics do not allow a nucleon to decompose spontaneously due to preserving the number of baryons. Its life is approximately 10 35 years, so it is considered eternal at a practical level.įrom the point of view of the standard model of particle physics, they are stable particles. Solar wind: it is a stream of charged particles released from the Sun's upper atmosphere. The cosmic rays, which are subatomic particles coming from space with a very high kinetic energy. It is a fluid state similar to the gaseous state but in which a certain proportion of its particles are electrically charged (ionized) and do not possess an electromagnetic balance. Plasmas: they are the fourth state of aggregation of the matter. All other types of atoms are made up of two or more protons and different numbers of neutrons. These two hydrogen isotopes are used as nuclear fuel in nuclear fusion reactions. For example, the nuclei of the heavy hydrogen isotopes ( deuterium and tritium) contain a proton and one or two neutrons, respectively. However, hydrogen has other isotopes that do contain neutrons. The only exception where it forms an atomic nucleus without any neutrons is the nucleus of an ordinary hydrogen atom - the most abundant nuclide in the universe. Protons and neutrons are the smaller particles belonging to the nucleus of an atom, so they are also called nucleons. These atomic sub-particles are present in atomic nuclei, generally attached to neutrons by a strong nuclear force. This structure means that it is a baryon (a subtype of particles called hadrons). It is made up of three gluon-bound particles, two up quarks and one down quark. The proton is not an elementary particle but a compound particle. The chemical elements are ranked in the periodic table of the elements accordingly to the atomic number. The number of this sub-particle in the atomic nucleus determines the atomic number. Just like going to South doesn't mean you've gone to a place less than nothing.A proton is a subatomic particle with a positive electrical charge found within the atomic nucleus of atoms. Here too, negatives are just keeping track of the direction. How can one do less than no work? This is not at all a 'mind-babbling' question because negative work is also some work. This 'greater' doesn't represent any 'improvement' in properties.Īnother great example is work-done: You might ask: One can either do some work or no work. Now, this 'greater' is not the same as 'greater' in everyday language. Even North could be represented by a '-' to keep track of sign.īut, if by greater, you mean 'more positive', then yes, the charge on a proton is $3.2\times10^$ greater than the charge on an electron only due to the sign convention (It could have easily been the other way around). If the result is a negative number, then you end up at South. For example, go 5 units North, then 3 units South, you end up at $5-3=2units$ North. But tracking South by a minus sign helps us in determining what is the end result or the effect. $0$ represents nothing and anything can't possibly be below nothing.Īsk yourself this: Is 5 units North 'greater than' 3 units South. But this 'less' is not the the same 'less' that we use in everyday world. People often consider that negatives are less than 0. Or negatives represent something which has the opposite effect of the positive quantity. In most cases, negatives just represent the opposite direction. Negative doesn't necessarily mean less, especially in Physics.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
